How Does Vivitrol Work? Breaking the Opioid Cycle
Article Contents
Opioid addiction is an epidemic steadily sweeping its way across America. Access to effective treatment is particularly important, as addiction is a complex, chronic disease – affecting people both psychologically and physically. From a first dabble with drugs, to a full-blown addiction, opioids such as heroin can quickly take hold of a person. Luckily, Vivitrol can help prevent relapse, but how does Vivitrol work?
People do recover from opioid addiction, but to do so successfully, they also need to deal with other issues present in their lives. Opioid addiction is not easy to escape from, but there are effective treatments available to make a life of sobriety possible.
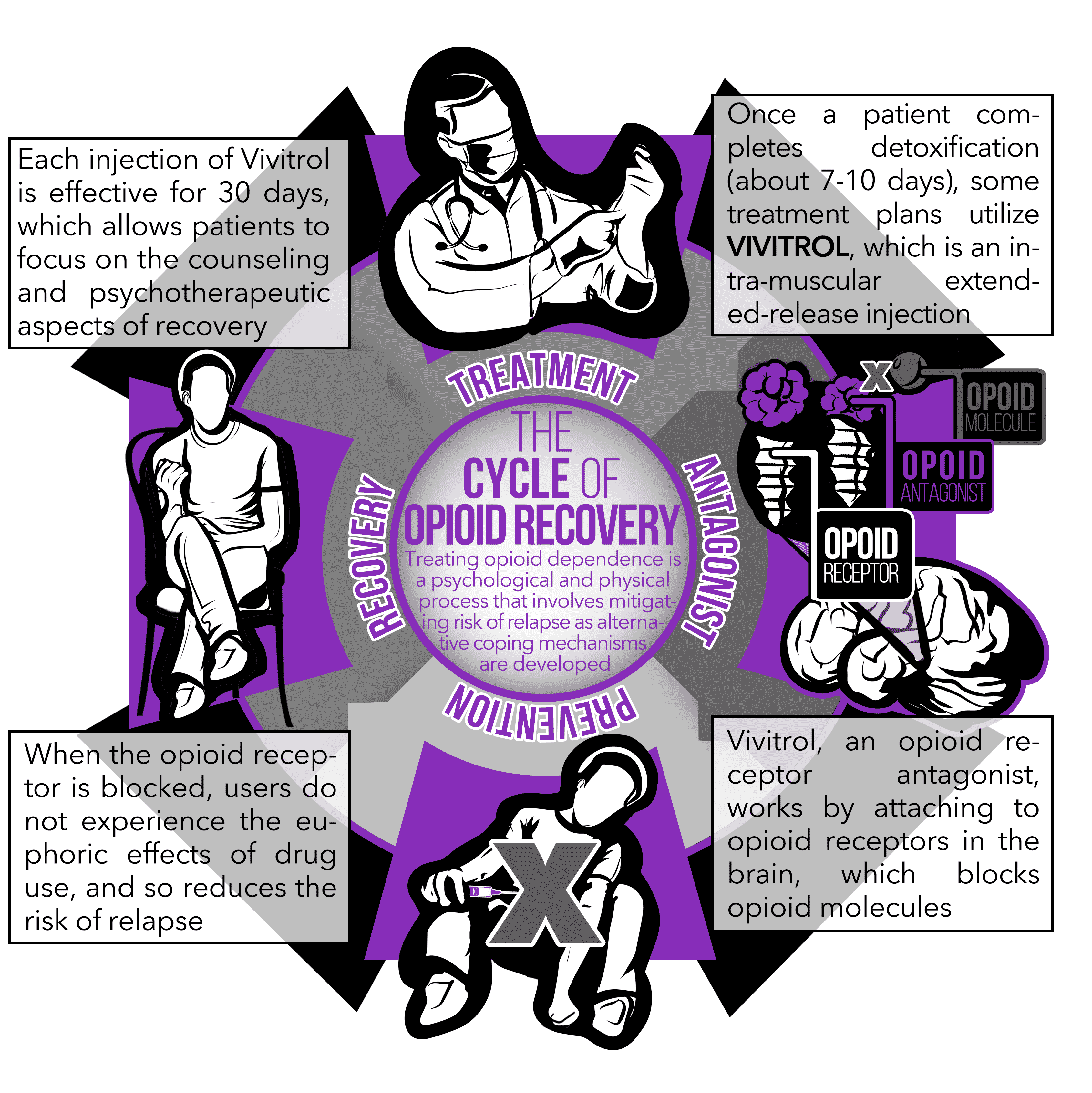
One such treatment and medication which can make a difference is Vivitrol. In October 2010, the FDA approved Vivitrol for the prevention of relapse following opioid detoxification.4 Vivitrol is the first and only non-narcotic, non-addictive medication available in a monthly injectable form approved for the treatment of opioid dependence. Vivitrol treatment may form part of a comprehensive management program, with the inclusion of psychosocial support.
How does Vivitrol work?
How does the Vivitrol shot work? Vivitrol, the trade name for an injectable formulation of naltrexone, works by attaching itself to opioid receptors in the brain, and consequently blocking them.5 In other words, it is known as an opioid receptor antagonist. This means that other opioids, such as heroin, are blocked from exerting their euphoric effects on the brain.
In order to improve patient adherence, naltrexone has been made available as an intra-muscular extended-release injection.5 Vivitrol was thus born. The single injection lasts for 30 days, unlike the alternative form of daily naltrexone pills. Naltrexone pills, in comparison, had limited efficacy because of the daily dosage required and subsequent poor compliance.4
Vivitrol can also work for the treatment of alcohol dependence. Johnson describes how Vivitrol has demonstrated efficacy with regards to decreasing heavy drinking in alcohol-dependent individuals2.
The body must be opioid-free for 7-10 days (and alcohol for at least 24 hours) prior to the administration of Vivitrol. Otherwise, severe withdrawal symptoms may be precipitated4. Once Vivitrol is administered, it isn’t possible to “get high” from heroin for a period of 30 days. For those battling addiction, that seems like a long time to commit – at least while the medicine remains within the body. One of the biggest hurdles, therefore, is the willingness of the person to surrender their habit.
Great excitement revolves around how it is administered; as an infrequent monthly shot. Also, unlike suboxone and methadone, Vivitrol cannot be misused or abused, and there is no illegal market for it.4
Adverse effects
Vivitrol isn’t suitable for all. There are significant risks from this treatment, including severe reaction at the injection site and severe opioid withdrawal. Other non-specific side effects may include nausea, headaches, and dizziness.1
If someone relapses and starts to use opioid drugs again, they will not experience a “high” since the antagonist is blocking the opioid receptors. Reusing opioid drugs after stopping Vivitrol thus carries an overdose risk as the user attempts a higher dose in order to achieve euphoria.1
However, the important take-home is that studies have failed to demonstrate overdose events, suicide attempts or deaths during Vivitrol trials.4
The future of Vivitrol
Despite the rising buzz around Vivitrol, how Vivitrol works, and its efficacy, the drug can’t necessarily be seen as a magic bullet for treating opioid users. It is an adjunct to treatment but not a cure for addiction. Regardless, receiving a monthly shot can be extremely helpful to those who wrestle daily with the decision to stay clean.
It was found to be more difficult to initiate patients on Vivitrol rather than buprenorphine-naloxone. Because of this, the overall relapse rate was negatively affected. However, once successfully initiated, both treatment options were equally safe and effective.3
Vivitrol is a monthly treatment, but it requires initial abstinence; the kind that is unlikely to be sustained out of a detox center. Furthermore, if you stop taking it, and you haven’t received help with coping strategies, you may relapse. After all, this treatment is only part of a recovery plan, not the sole solution. A person has to stay in the rehabilitation facility (or stay sober) for any treatment to be helpful, but Vivitrol can make it possible for people to continue the recovery process on good footing.
For treatment to be successful, rehabilitation programs must combine counseling, psychotherapeutic work, and medication in order to help a person move forward in their recovery process.
References
- Armand, W. (2016). A monthly shot for opioid addiction: An option for some. Retrieved March 15, 2019 from https://www.health.harvard.edu/blog/a-monthly-shot-for-opioid-addiction-an-option-for-some-201604019364
- Johnson, B. A. (2007). Naltrexone long-acting formulation in the treatment of alcohol dependence. Therapeutics and clinical risk management, 3(5), 741-9.
- Lee, J.D., Nunes, E.V., Novo, P., Bachrach, K., Bailey, G.L., Bhatt, S., . . . Rotrosen, J. (2018). Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicenter, open-label, randomised controlled trial. Lancet, 27;391(10118):309-318.
- Ndegwa S., Pant S., Pohar S., & Mierzwinski-Urban, M. (2017). Injectable Extended-Release Naltrexone to Treat Opioid Use Disorder. [Bulletin]. CADTH Issues in Emerging Health Technologies. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health.
- Syed, Y.Y. & Keating, G.M. (2013). Extended-Release Intramuscular Naltrexone (VIVITROL): A Review of Its Use in the Prevention of Relapse to Opioid Dependence in Detoxified Patients. CNS Drugs, 27(10): 851-861.